SUPERNUS PHARMACEUTICALS (SUPN)·Q4 2025 Earnings Summary
Supernus Posts Record Revenue as Growth Products Surge 45%, Stock Jumps 9%
February 24, 2026 · by Fintool AI Agent

Supernus Pharmaceuticals delivered record quarterly revenue of $211.6 million in Q4 2025, up 21% year-over-year, driven by 45% growth in its four key growth products . The CNS-focused biopharma announced FY2026 revenue guidance of $840-870 million, representing 17-21% growth, as ONAPGO new patient initiation resumed following supply constraints .
The stock jumped 5.1% to $53.30 during regular trading and surged another 4% aftermarket to $55.39, as investors looked past GAAP losses driven by one-time acquisition costs from the July 2025 Sage Therapeutics deal.
Did Supernus Beat Earnings?
Revenue: Beat by 6.6% — Q4 revenue of $211.6M exceeded the $180M consensus estimate significantly . Total revenues for FY2025 reached a record $719.0M, up 9% year-over-year .
EPS: Missed by 25.6% — Normalized EPS of $0.61 fell short of the $0.82 consensus. The miss was largely driven by $72.9 million in acquisition-related costs from the Sage Therapeutics acquisition, including $25M in accelerated equity compensation . On a GAAP basis, Supernus reported a net loss of $4.1M (-$0.07 per diluted share) compared to net income of $15.3M ($0.27 per share) in Q4 2024 .
Adjusted Operating Earnings: In Line — Adjusted operating earnings of $48.5M were essentially flat versus $48.3M in Q4 2024, demonstrating underlying business stability despite acquisition noise .
How Did the Stock React?
SUPN shares rallied sharply following the earnings release:
- Regular session: +5.1% to $53.30 (from $50.69 close)
- Aftermarket: Additional +3.9% to $55.39
- Total move: ~9% gain from prior close
The stock is now trading near its 52-week high of $57.65, having more than doubled from its 52-week low of $29.16. The positive reaction suggests the market is focused on the growth product trajectory and 2026 guidance rather than the GAAP losses driven by one-time acquisition costs.
What Changed From Last Quarter?
Key changes from Q3 2025:
-
ONAPGO supply resolved — New patient initiation has resumed in Q1 2026 after being paused due to supply constraints. A second supplier is expected to begin supplying ONAPGO in 2027 .
-
ZURZUVAE ramping — Collaboration revenue from ZURZUVAE reached $32.8M in Q4, representing Supernus's 50% share of Biogen's net revenues. Q4 U.S. sales increased 187% YoY and 19% sequentially .
-
Acquisition costs behind — The Sage acquisition closed in July 2025 with $72.9M in one-time costs largely recognized in 2025. This noise should clear in 2026 .
-
Legacy erosion accelerating — Trokendi XR (-43% YoY) and Oxtellar XR (-48% YoY) continue declining due to generic competition, but now represent only 7% of total revenue .
What's Driving Growth? Product Portfolio Deep Dive
The growth products story is the key to understanding Supernus's trajectory. Combined revenue from Qelbree, GOCOVRI, ZURZUVAE, and ONAPGO reached $161.3M in Q4 2025, up 45% year-over-year .
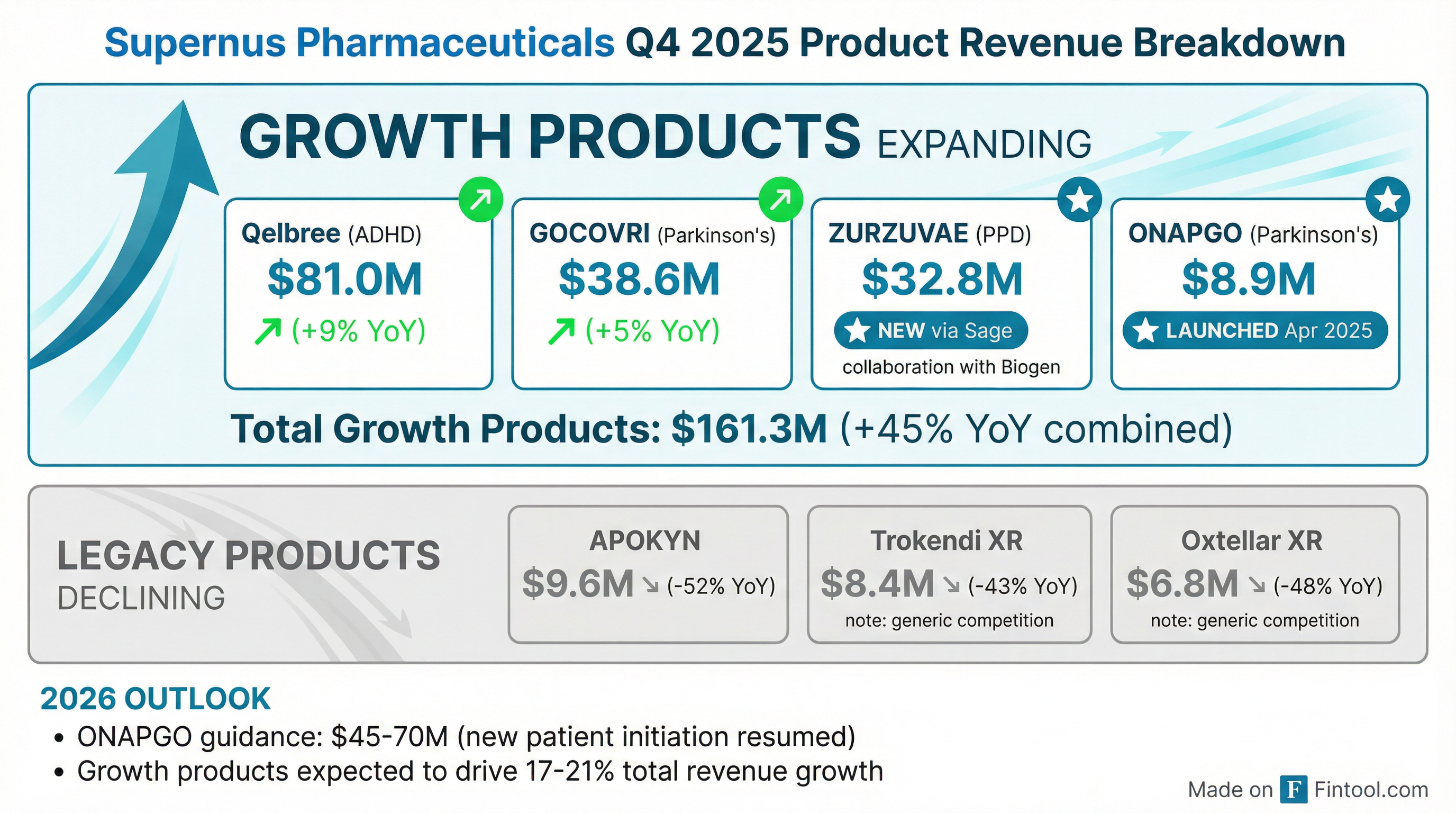
Qelbree continues as the flagship, with total IQVIA prescriptions up 18% year-over-year in Q4 . The 9% revenue growth was partially offset by an annual gross-to-net deduction reflected in Q4.
ZURZUVAE, acquired through Sage, represents a major new revenue stream for postpartum depression treatment. Total prescriptions increased over 150% in 2025 versus 2024 .
ONAPGO launched in April 2025 with over 1,800 enrollment forms from 540+ prescribers since launch. The supply resumption positions this as a key 2026 growth driver .
What Did Management Guide?
Supernus provided robust FY2026 guidance reflecting confidence in the growth product portfolio:
The guidance implies:
- ONAPGO as the key swing factor with $45-70M guidance range representing significant acceleration with supply constraints resolved
- Legacy products (Trokendi XR + Oxtellar XR) expected at $40-50M, continuing controlled decline
- R&D + SG&A expenses guided at $620-650M
Pipeline Update: What's Coming?
Supernus has several CNS pipeline programs advancing:
SPN-817 (Epilepsy) — Phase 2b study ongoing in ~258 adults with treatment-resistant focal seizures. Novel first-in-class AChE inhibitor .
SPN-820 (Depression) — Follow-on Phase 2b initiated in ~200 adults with major depressive disorder. Novel mechanism targeting mTORC1 mediated synaptic function .
SPN-443 (ADHD) — Novel stimulant with Phase 1 study expected in second half of 2026 .
Balance Sheet and Capital Position
Cash decreased $144.9M year-over-year primarily due to funding the Sage acquisition, partially offset by operating cash flow . The company maintains a debt-free balance sheet with ~$309M in liquidity to fund operations and pipeline advancement.
Key Risks and Concerns
- ONAPGO supply dependency — While a second supplier is expected in 2027, near-term supply relies on a single source
- Legacy product erosion — Trokendi XR and Oxtellar XR facing generic competition with combined revenue declining ~40% YoY
- Integration risk — Sage acquisition integration ongoing with potential for additional costs or disruptions
- Competitive pressure — ADHD and Parkinson's markets face increasing competition
The Bottom Line
Supernus delivered a strong Q4 with record revenue and demonstrated that the growth product strategy is working. The 45% growth in the core product portfolio more than offsets legacy erosion, and the Sage acquisition adds meaningful new revenue streams in ZURZUVAE.
The GAAP losses are noise from one-time acquisition costs — underlying profitability remains stable with adjusted operating earnings holding flat year-over-year. With ONAPGO supply constraints resolved and guidance calling for 17-21% revenue growth in 2026, the stock's 9% post-earnings rally appears justified.
Key metrics to watch going forward:
- ONAPGO prescription trends following supply resumption
- ZURZUVAE collaboration revenue trajectory
- Qelbree market share in ADHD
- Pipeline readouts for SPN-817 and SPN-820
Conference call held February 24, 2026 at 4:30 PM ET. Replay available at supernus.com/investors